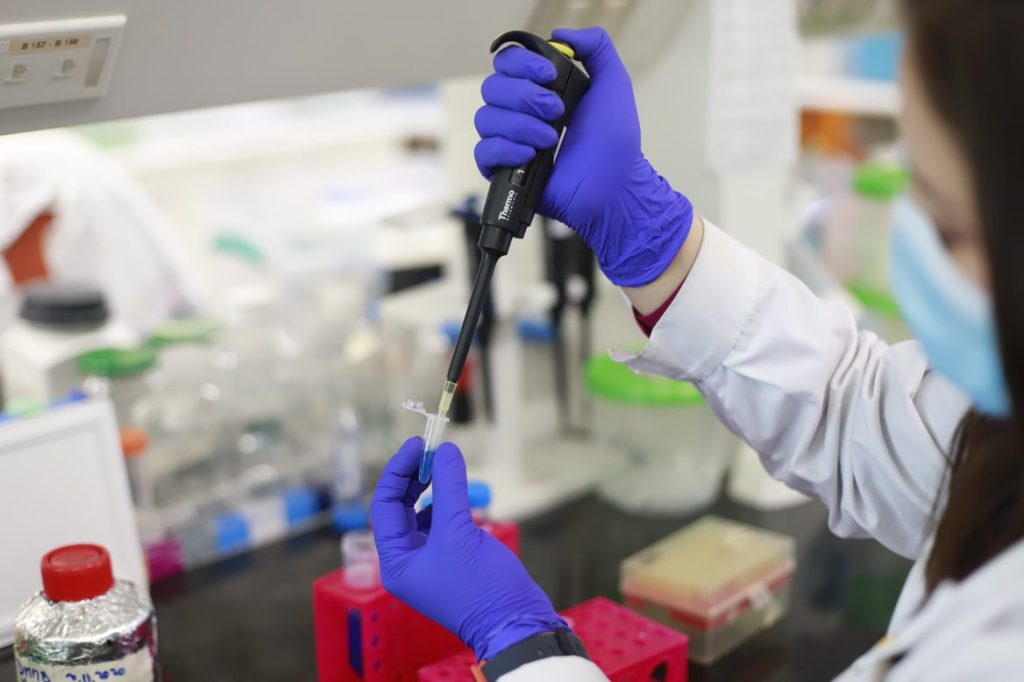
Clinical Trials
At IMPACT we are committed to advancing toward quality, accessible and innovative healthcare, bringing scientific advances to the clinic and bringing their results closer to those who need them most: people.
Address
Avenida La Plaza N°2501, Las Condes. Santiago de Chile
Contact
Write to us
What is a clinical trial?
Why participate?
Who can participate?
A clinical trial is an investigation conducted on human subjects aimed at determining or verifying the effects, efficacy and safety of the investigational product.
Clinical trials offer a therapeutic opportunity to patients who have exhausted all standard treatments. They also provide an opportunity to participate in medical research and contribute to the advancement of science.
To participate you must meet a number of inclusion criteria. Participation is completely voluntary and you may decline to participate at any time during the trial.
Phases of clinical trials
Phase I
It is performed on a small number of volunteers The primary focus is to assess the safety of the investigational product and side effects.
Phase II
The objective is to evaluate efficacy and further safety data. Search for the optimal dose of the product. It is performed on a smaller group of volunteers, but larger than in Phase I.
Phase III
The research is being conducted on a large group of patients worldwide. It is the last link in testing a new treatment in humans. When Phase III studies have been completed, and the treatment can be shown to be safe and effective under certain conditions, it is ready for regulatory approval to go to market. The regulatory authorities approve its release to the market.
Phase IV
Post-marketing studies are conducted in search of more data on safety, efficacy and better uses.
Clinical trials
Status: OPEN
The treatment consists of a new formulation of a Cell Therapy developed from Stem Cells derived from umbilical cord blood. The objective of the present study is to use the same type of cells, but in a formulation that can be frozen for wider distribution and direct use after thawing, without the need for expensive devices or equipment.
We have previously demonstrated that the use of these cells-also called “Mesenchymal Stromal Cells” or MSCs-, are effective for the treatment of osteoarthritis of the knee.
Contact us: [email protected]
Status: OPEN
Phase II clinical trial to evaluate the dose-response and efficacy of umbilical cord-derived mesenchymal stromal cells (MSCs) in severe renal systemic lupus erythematosus (SLE).
Contact: [email protected]
Status: OPEN
Study of patients with depression and bipolarity for the development of a multiparametric algorithm for the detection of mood disorders.
Contact: [email protected]
Status: CLOSED
Phase I clinical trial to evaluate the clinical safety of CelliStemOA-sEV.
Status: CLOSED
Cohort study of healthy newborns for gut microbiota characterization.
Status: CLOSED
Cohort study of pregnant women, 38-40 weeks gestation for the evaluation of biomarkers in biological fluids in patients with and without labor.
Status: OPEN
Cohort study of women of childbearing age, without use of hormonal contraceptive method in search of pregnancy, for the identification of molecular markers in pre-conception stage.
Contact:
Status: CLOSED
Cohort study of pregnant women between 35-37 weeks gestation for the evaluation of late fetal growth screening parameters.
Status: CLOSED
Case study of women with pregnancy diagnosed with trisomy 21 (Down syndrome).
Status: CLOSED
Cohort study of pregnant women between 11-14 weeks gestation for early detection of gestational diabetes.
Status: OPEN
Cohort study of women of childbearing age, without hormonal contraceptive use for the development of a multiparametric algorithm for the early detection of endometriosis.
Contact:
Status: CLOSED
Evaluation of women with early deliveries to assess the impact of mitochondrial transfer in cord blood transplantation and its relevance to GVHD disease.
Status: OPEN
Controlled Phase I clinical trial to evaluate the safety of Cellistem®ER-Sev in patients with apical periodontitis for dental pulp regeneration.
Contact: